
Efficacy refers to how well the vaccine is meeting its desired goals in a clinical trial (e.g.Immunogenicity refers to how well a vaccine is at getting the immune system to respond to the vaccine. For a vaccine to work well, it needs to be recognized by the immune system and generate a response.Phase II: looks at the immunogenicity and efficacy of the vaccine and determines the proper dose and route of administration.Phase I: checks the safety of the vaccine in humans (typically the smallest and shortest trial).The FDA regulates vaccine candidates throughout all phases of clinical trials. Each phase of the trial has a different purpose, and typically the size and length of the trial increases through the phases. The clinical phases are subsequent trials of vaccines in people with the goal to test the safety and efficacy of the vaccines. An Investigative New Drug (IND) application using the pre-clinical data must be submitted and approved by the FDA before moving to clinical trials in people. Once a promising candidate has been found in the R&D phase, animal models are used to test the safety and immunogenicity of the vaccine. SARS-CoV-2/COVID-19) and spend 2-4 years conducting research and developing a vaccine candidate. Scientists will come up with a vaccine approach to a target (e.g. Standard ProcessĮvery vaccine candidate begins with research. The COVID-19 pandemic created the need for this “fast-track” to be implemented. It’s important to note that the FDA was working on modernizing clinical trials, including creating guidance for adaptive designs, back in 2018. Additionally, due to the COVID-19 pandemic, the FDA also was granting Emergency Use Authorization (EUA) so that the vaccines could be administered to the public more quickly. During the COVID-19 pandemic, the FDA utilized a Fast-Track method that helped vaccines be tested more quickly without compromising on safety goals. There is a Standard Process that researchers will use to go through clinical trials and vaccine approval. 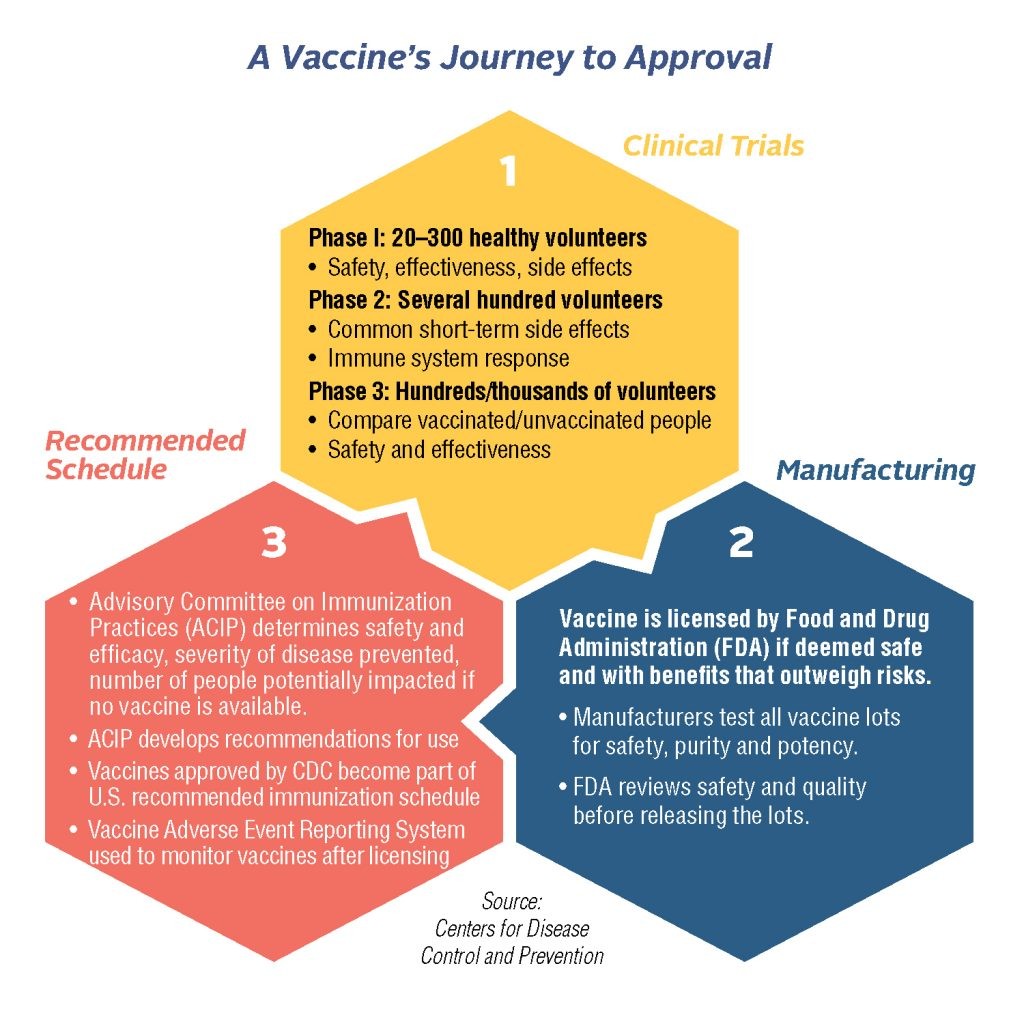
Public Health Service Corps deploymentĪpproximately 364 CDER Commissioned Corps officers fulfilled 623 deployment requests in support of the COVID-19 mission Engagement with stakeholdersĬDER fielded 23,515 COVID-19 drug related inquiries from the general public, including health care providers, consumers and manufacturersįor more information, please visit vaccines distributed in the USA are under regulation of the FDA. Published a list with more than 250 listings of hand sanitizers consumers should not use, including those containing potentially dangerous contaminants Shortage mitigation activitiesĬontinued outreach to more than 180 manufacturers relating to manufacturing capacity and supply chain for both COVID-19 and non-COVID-19 treatments Surveillance and EpidemiologyĮxamined and analyzed data across at least 15 data sources, including 55,000 adverse event reports to monitor drug safety and medication errors U.S. Issued 17 warning letters to operators of websites that sell unapproved and misbranded COVID-19 products Hand sanitizer Took action against sellers of fraudulent products for the treatment or prevention of COVID-19 and issued more than 120 warning letters Internet pharmacies Generic Drug Approvals (Abbreviated New Drug Applications)Īpproved 1,000+ original and supplemental generic applications for COVID-19 related treatments and supportive therapies Addressing fraud Drug Supply Chain Security Act requirements.Prescription Drug Marketing Act requirements for distributing prescription drug samples.Certain drugs compounded for hospitalized patients with COVID-19.Proactively issued temporary policies to address the pandemic and provide regulatory flexibility on: 11 treatments currently authorized for emergency use.640+ drug development programs in planning stages.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
